The Genomics Age of Cancer Screening: Earlier Cancer Detection

Forward
Researchers are unlikely to find the cure for cancer in a single therapy. In fact, the most efficient way to combat cancer may not be a cure at all, as “an ounce of prevention is worth a pound of cure”. This article aims to quantify how prevention could impact cancer mortality. Forthcoming articles will focus on earlier cancer detection from other angles: biology, morbidity, unit economics, and the total addressable market. Moreover, in the spirit of transparency, we will open our model(s) for comments, questions, and suggestions, all of which will be incorporated into a comprehensive whitepaper early next year.
Why Earlier Detection?
Cancer is easier to treat when diagnosed early. All solid tumors start as benign lesions called neoplasia that advance from local, to regional, and finally to distant or metastatic cancer. As cancer progresses, the number of treatment options shrinks. Surgeons lose the ability to remove tumors completely and chemotherapies become ineffective. Unfortunately for many patients, symptoms don’t present until their cancers have spread. In lung, pancreatic, and ovarian cancers, for example, most tumors are metastatic by the time they are diagnosed. Over time, metastatic cancers account for a minority of new cases but cause the majority of deaths, as shown below.
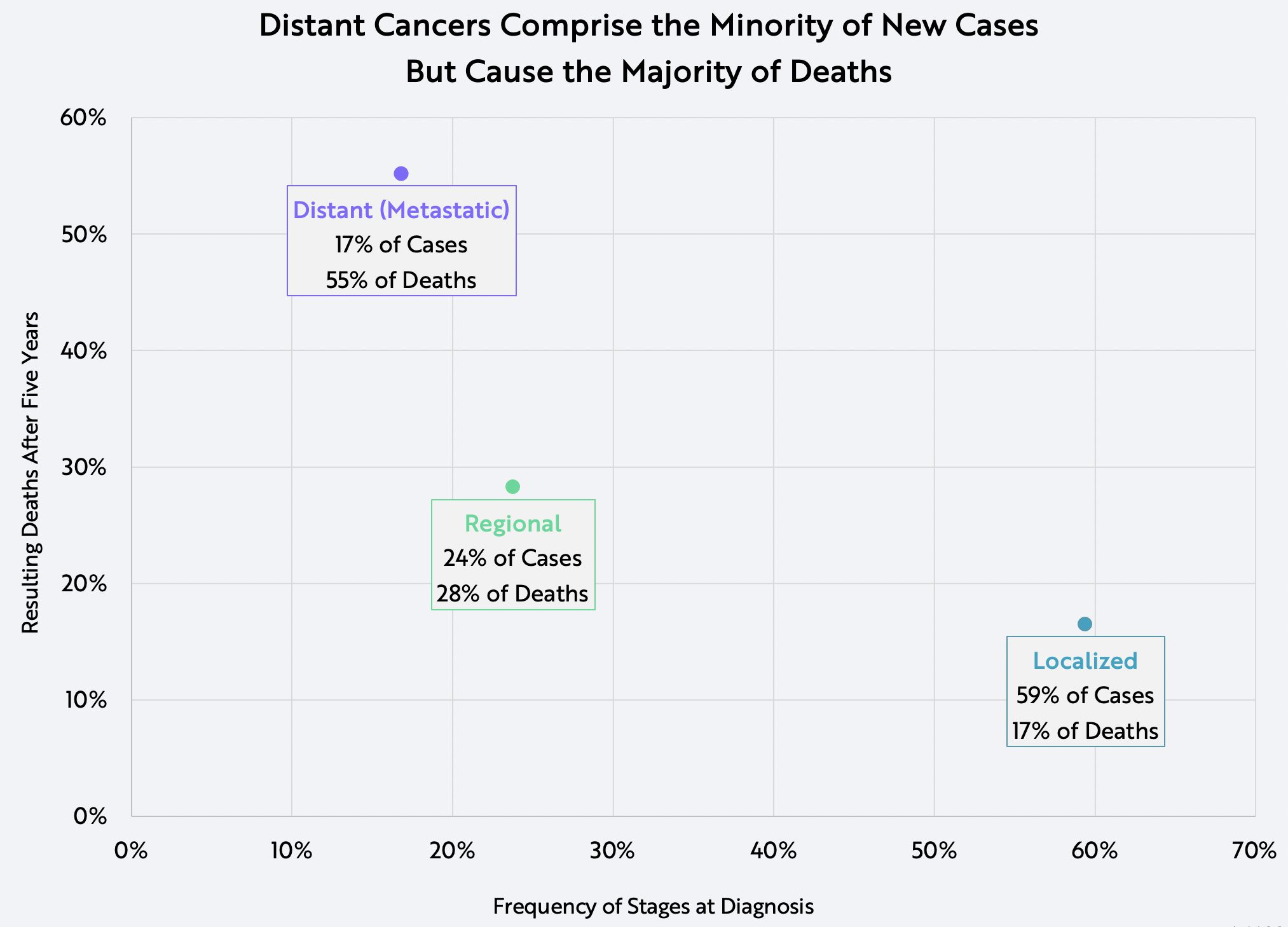
Source: ARK Investment Management LLC, 2020 data sourced from: SEER. “SEER*Explorer.” Surveillance, Epidemiology, and End Results Program, 2020, seer.cancer.gov/explorer/.
The rationale for screening is that cancer follows a predictable path—from local and treatable to metastatic and lethal. Conceptually, earlier detection maximizes access to available therapies and interventions before the cancer becomes intractable. Though ARK assumes treatments for late-stage cancers will improve, those drugs are unlikely to cure cancer and could be too specialized, benefiting only a small number of patients. The design of a robust tool to detect cancer at an early stage in large swaths of the global population will be critical to reducing cancer mortality rates.
The Current Screening Standard
The United States Preventive Services Task Force (USPSTF), an independent consortium of physicians, medical scholars, and policymakers, publishes national cancer screening guidelines intermittently. Since its creation in 1984, the USPSTF has issued screening recommendations only for breast, cervical, and colorectal cancers. Without more formal guidance, physicians often screen for liver, lung, and prostate cancers as well.
Today, nearly all screening technologies are half a century old and, in the context of recent scientific breakthroughs, flawed. Medical imaging techniques like mammography and low-dose CT (LDCT) scanning are highly sensitive, for example, but suffer from poor specificity—generating false positives, as defined below. Moreover, they are limited to certain sites, not to the whole body. Common proteomic biomarkers used to screen for tumors – including carcinoembryonic antigen (CEA), prostate specific antigen (PSA), and cancer antigen 125 (CA-125) – also are limited in their sensitivity-specificity profiles as well as the number of cancers they can detect.
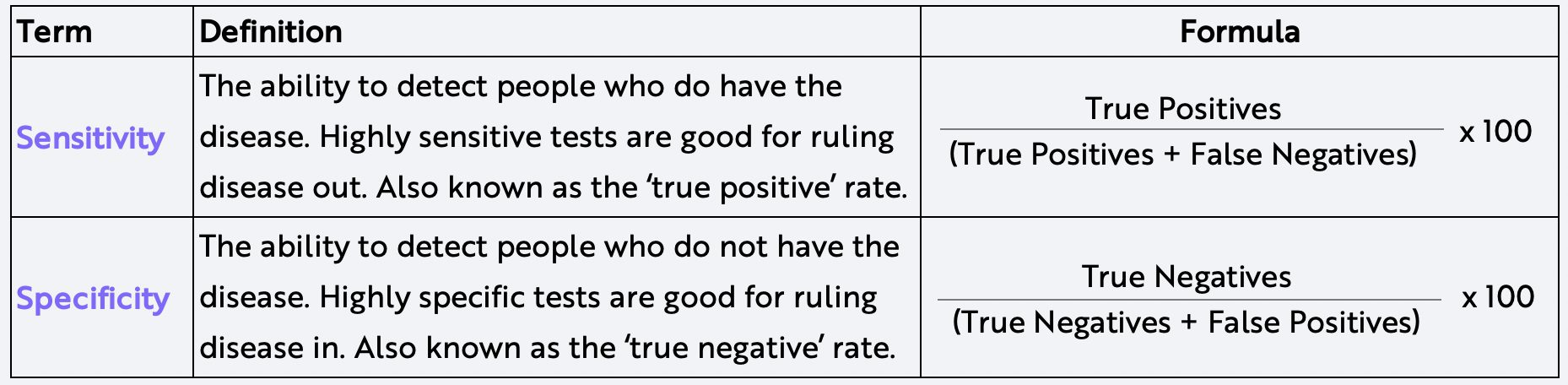
Source: ARK Investment Management LLC, 2020
Because of limited accuracy, poor resolution, and misperceptions, many physicians and patients are hesitant to comply with national cancer screening guidelines. Some patients eligible for colonoscopies – semi-invasive tests to detect colorectal cancer – avoid screening out of fear that the test itself will cause harm or generate false results. Consequently, many professionals in the medical community are pessimistic about the prospects for cancer screening.
We agree that legacy screening tools are not the answer to earlier cancer detection. Instead, liquid biopsies—molecular tests incorporating decades of genomic insight, cutting-edge machine learning, and synthetic biology—are likely to move to the frontline of cancer screening now that the technologies have advanced and their costs have dropped to critical thresholds.
Cancer Screening in the Post-Genome Era
Concluded in 2003, the Human Genome Project marked the beginning of the post-genome era. In our view, genomics is the foundation of personalized medicine, enabling treatments based on genetic mutations unique to each individual.
Approved by the FDA in 2014, Exact Sciences’ (EXAS) Cologuard launched colorectal cancer (CRC) screening in the post-genome era. Shipped to patients’ homes, Cologuard surfaces microscopic genetic mutations derived from tumors in stool. Also non-invasive, modern liquid biopsies can detect numerous cancers in blood. In these early days, we think multi-cancer screening tests should focus on a narrower set of cancers, like pancreatic, that are difficult to identify early. As clinical evidence builds over time, however, these assays can expand to cover more cancer types.
Reduction of Cancer Mortality
Recall that cancer is a progressive disease. All solid tumors begin as benign neoplasia and progress at different rates towards incurable metastatic states. Adjusted for the incidence of different cancers, the weighted-average five-year mortality rate for localized cancers is only 11% while that for distant cancers is 76%, as shown below. In other words, 89% of patients with localized cancers survive for five years after diagnosis, well above the 24% with metastatic cancers who survive.
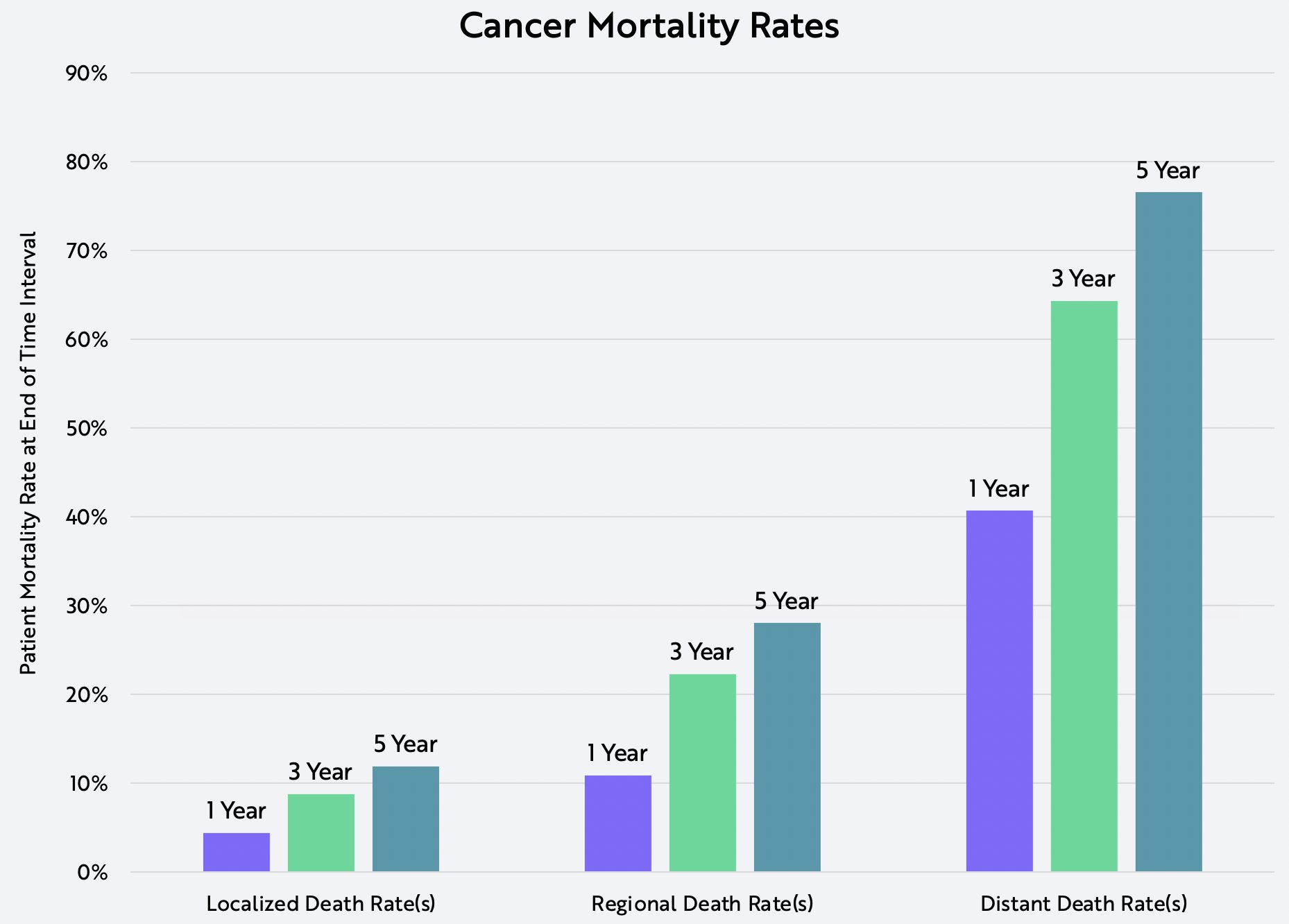
Source: ARK Investment Management LLC, 2020 data sourced from: SEER. “SEER*Explorer.” Surveillance, Epidemiology, and End Results Program, 2020, seer.cancer.gov/explorer/.
In our view, multi-cancer screening could reduce the death rate by shifting average diagnoses from distant towards localized cancers. We arrive at that conclusion after the following analysis. First, we select solid tumor types known to produce detectable quantities of circulating tumor DNA (ctDNA) and/or tumors already included in multi-cancer liquid biopsy screening studies. Our model incorporates both age- and tumor-specific incidence rates as well as stage-specific incidence and mortality statistics from the Surveillance, Epidemiology, and End Results (SEER) Cancer Database. While we expect assay sensitivity and specificity to rise as training data proliferates, GRAIL’s published performance on restricted cancers provides the current baseline (sensitivity = 0.67; specificity = 0.99). Finally, for our selected list of cancers, we assume that the frequency of each diagnosis stage is 70% localized, 25% regional, and 5% metastatic, as explained in the table below.

Source: ARK Investment Management LLC, 2020
With these inputs, we simulate the impact that multi-cancer liquid biopsy screening has on cancer-specific mortality. In the US, as shown below, after five years of national adoption, an annual screen that detects (a) cancers not covered by existing tests in (b) people older than 50 could lower the number of cancer deaths by 56,000 per year. Adding to the list of cancers covered by existing screening tests, multi-cancer liquid biopsies could lower the number of cancer deaths by 60,000 per year. Finally, including eligible patients older than 40, multi-cancer screening could prevent 66,000 cancer deaths per year, as shown below.
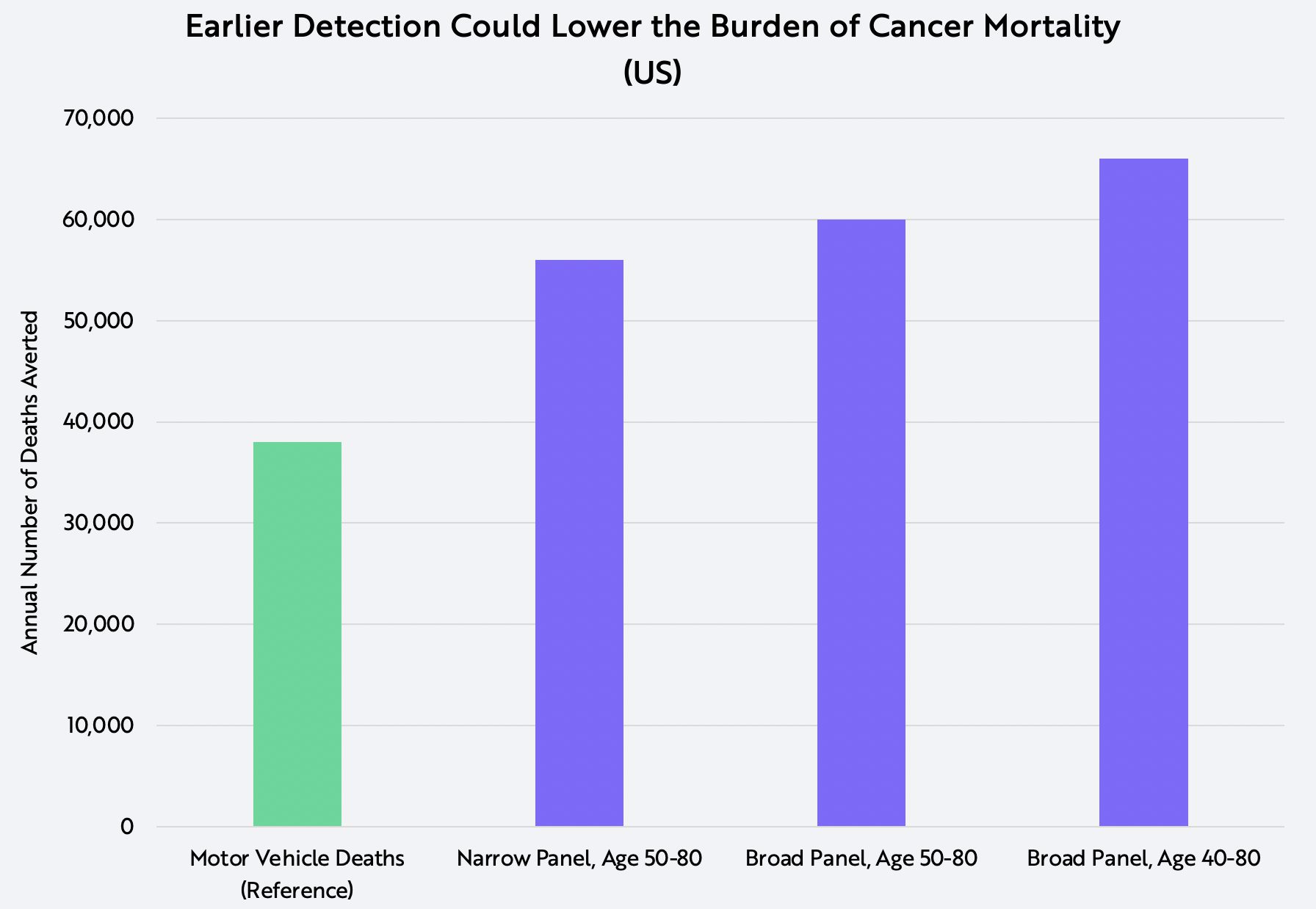
Source: ARK Investment Management LLC, 2020 data sourced from: SEER. “SEER*Explorer.” Surveillance, Epidemiology, and End Results Program, 2020, seer.cancer.gov/explorer/.
In the face of our analysis that multi-cancer liquid biopsy screening could lower cancer-specific deaths by 66,000 per year, we also understand the obstacles to widespread adoption in the near term. While some companies in the space may pursue near-term commercialization, we do not expect FDA approval nor reimbursement to occur until at least 2023. Without these prerequisites, widespread adoption simply is not possible. Additionally, companies will need to address issues such as lead-time bias, over- or unnecessary treatment, and the significant costs relative to the current standard of care.
In future blogs, we will elaborate on all of the aforementioned hurdles to widespread clinical adoption. To incorporate constructive criticism and other analyses for our forthcoming whitepaper on the impact of earlier cancer detection, we will open-source our cancer screening model(s) on GitHub in the coming weeks.

 Actively Managed Equity
Actively Managed Equity Overview: All Strategies
Overview: All Strategies Investor Resources
Investor Resources Indexed Equity
Indexed Equity Private Equity
Private Equity Digital Assets
Digital Assets Invest In The Future Today
Invest In The Future Today
 Take Advantage Of Market Inefficiencies
Take Advantage Of Market Inefficiencies
 Make The World A Better Place
Make The World A Better Place
 Articles
Articles Podcasts
Podcasts White Papers
White Papers Newsletters
Newsletters Videos
Videos Big Ideas 2024
Big Ideas 2024


